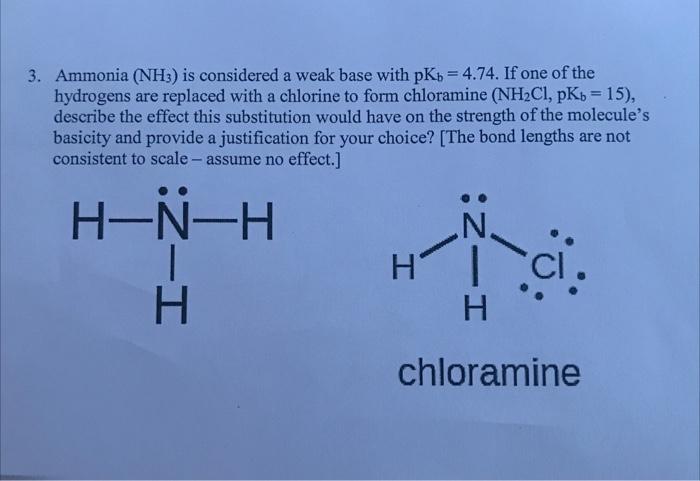
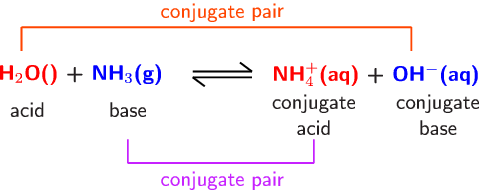
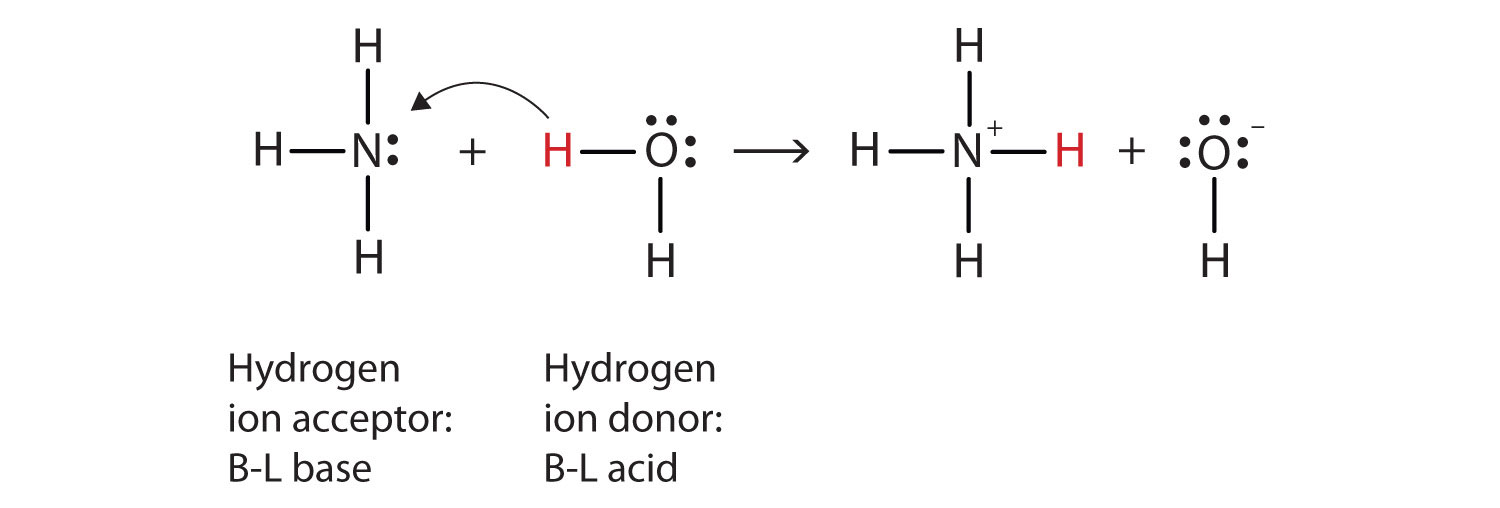
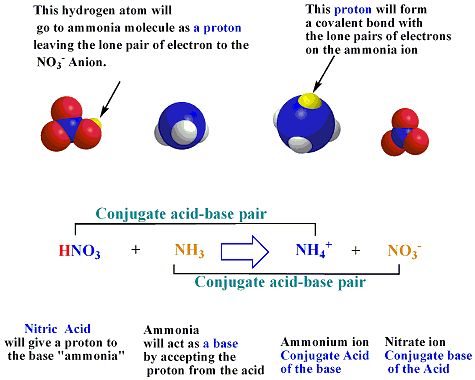
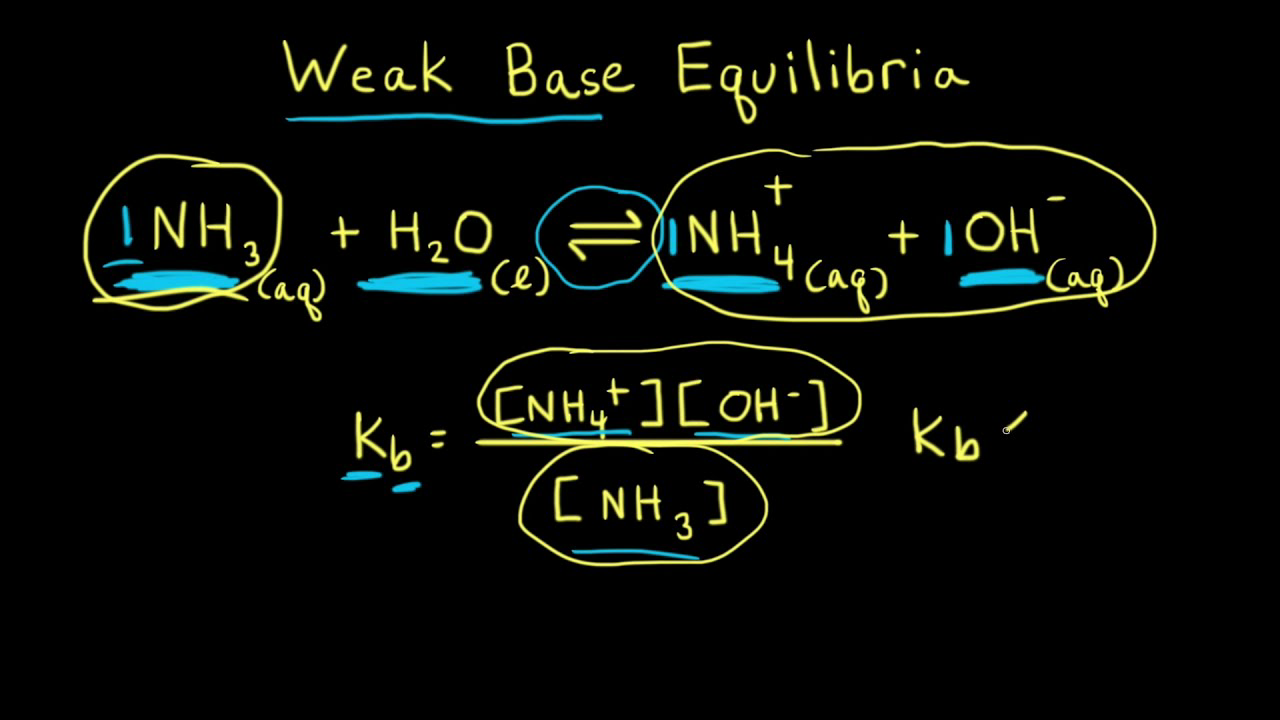redox - Why does ammonia act as an acid in only the first of these two reactions? - Chemistry Stack Exchange

a) Explain why ammonia acts as a weak base in water. (b) Write a balanced chemical equation for the reaction between ammonia and water. | Homework.Study.com

Question Video: Identifying the Lewis Acid in the Reaction of Ammonia with Boron Trifluoride | Nagwa
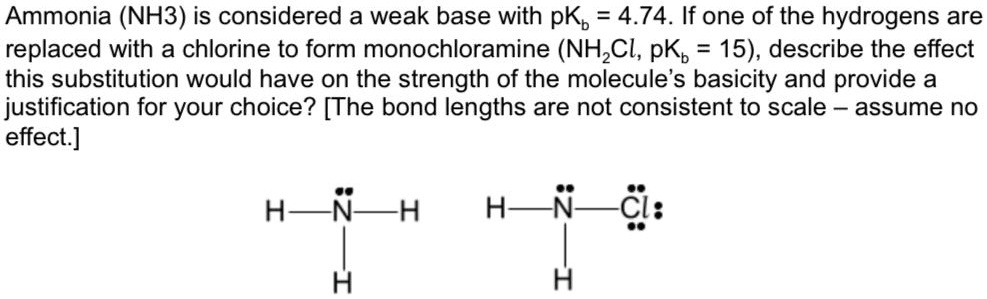
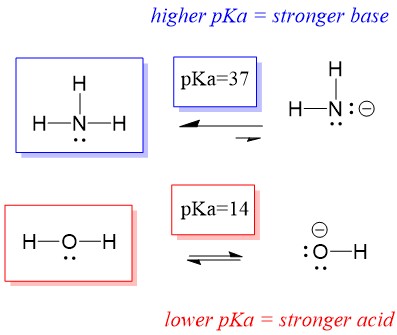







![Why does \\[N{H_3}\\] act as a Lewis base? Why does \\[N{H_3}\\] act as a Lewis base?](https://www.vedantu.com/question-sets/db470ea0-7477-412c-82d8-a4917a1132145281377342200076057.png)