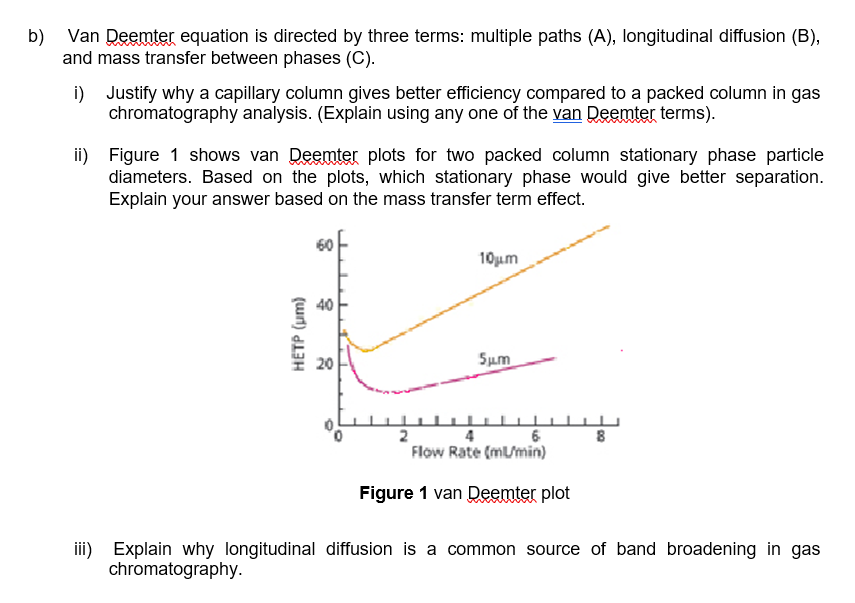
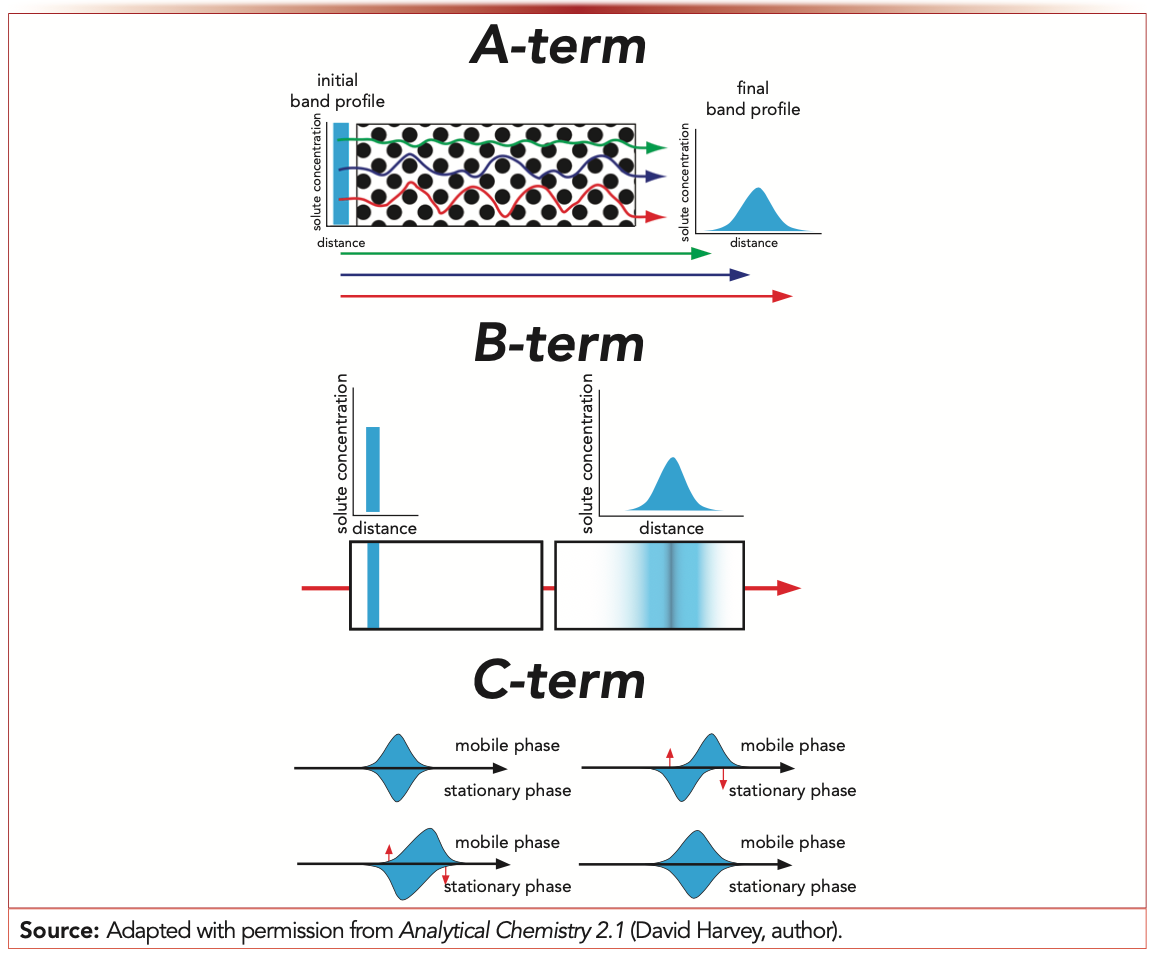
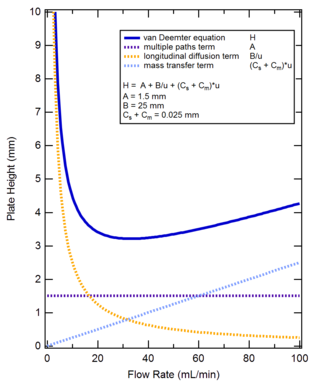
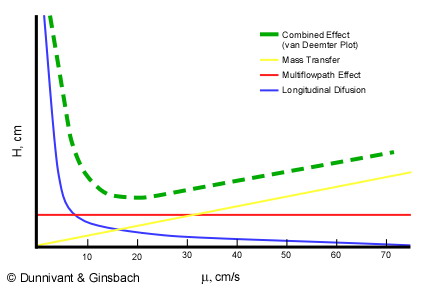
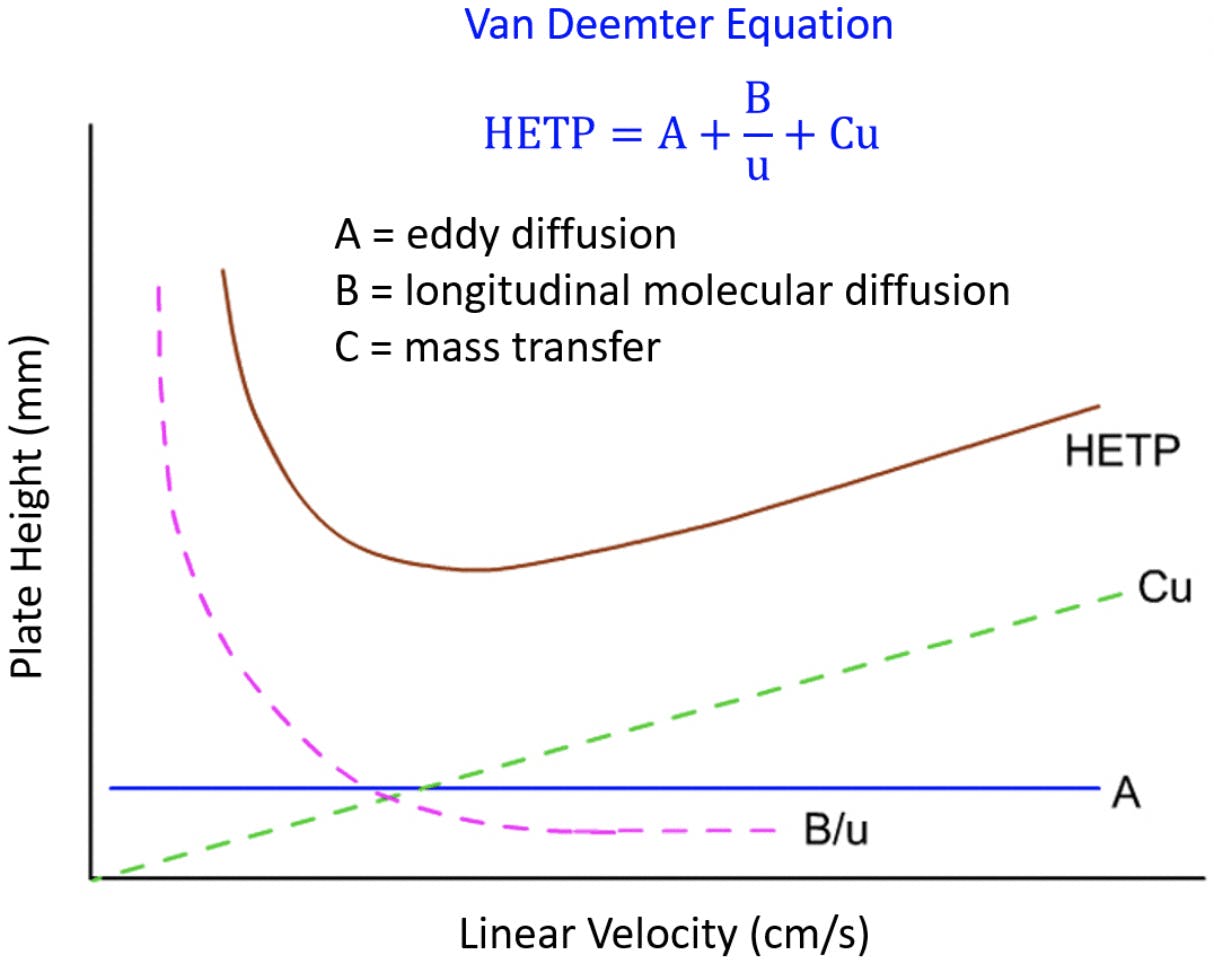
Evaluation of the van Deemter equation in terms of open‐ended flow to chromatography - Andersen - 2020 - Journal of Separation Science - Wiley Online Library

Investigation of the Separation Efficiency of Tube Radial Distribution Chromatography with Stationary Outer Phase Using the van Deemter Equation | SpringerLink

Evaluation of the van Deemter equation in terms of open‐ended flow to chromatography - Andersen - 2020 - Journal of Separation Science - Wiley Online Library
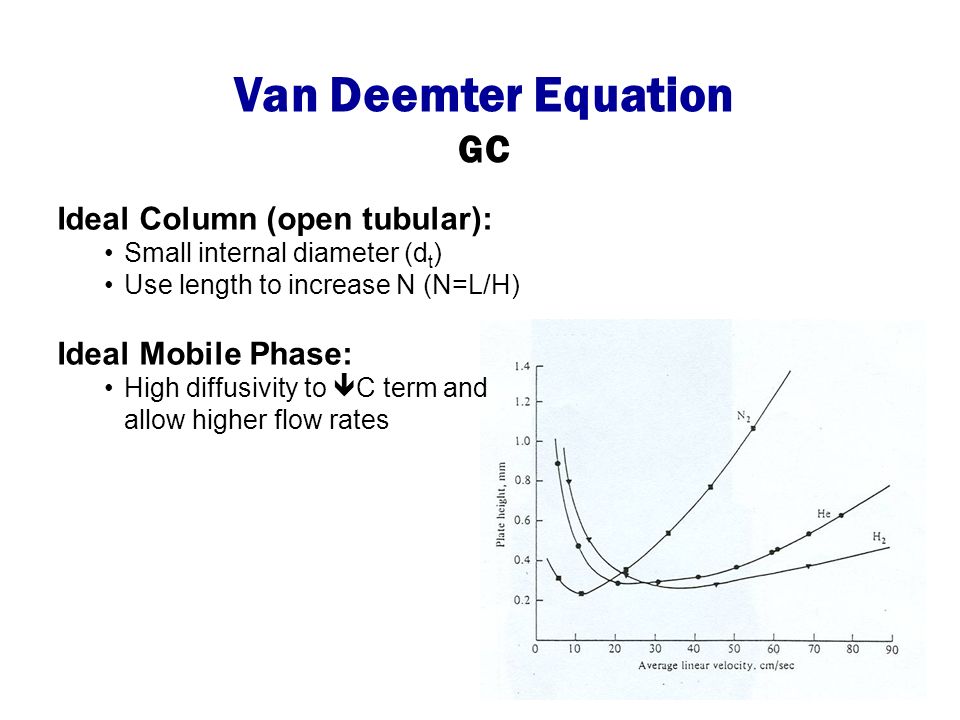
Lecture 8 Van Deemter Equation!. Resolution Describes how well 2 compounds are separated Rs = 1 4 N 1/2 ( -1) k' 1+k' ( ) efficiency selectivity retention. - ppt download