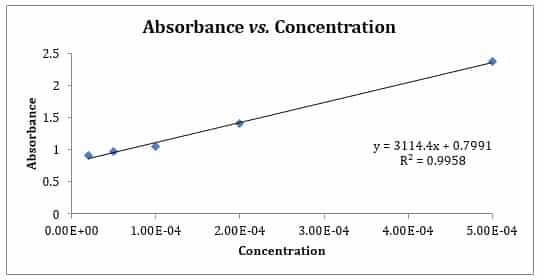
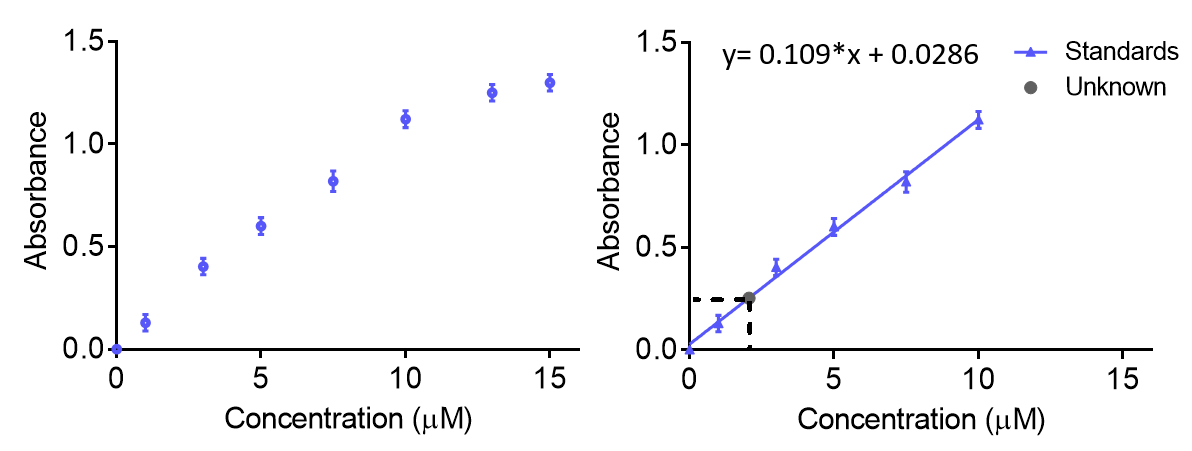
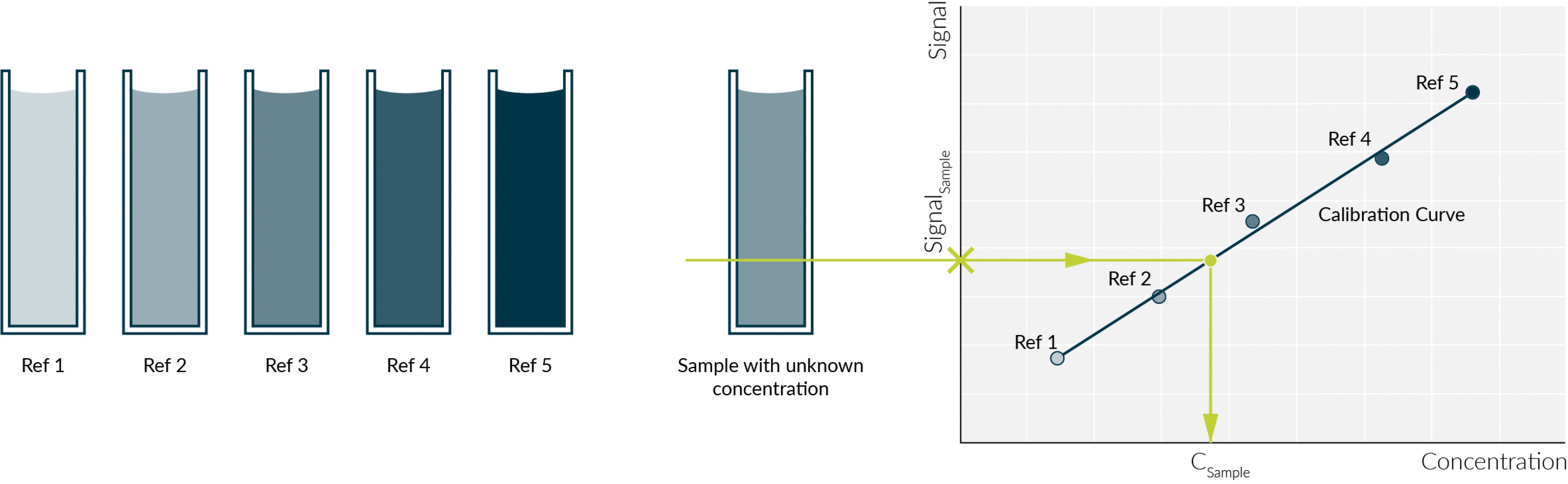
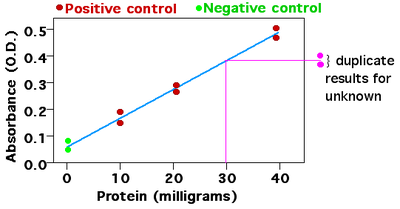
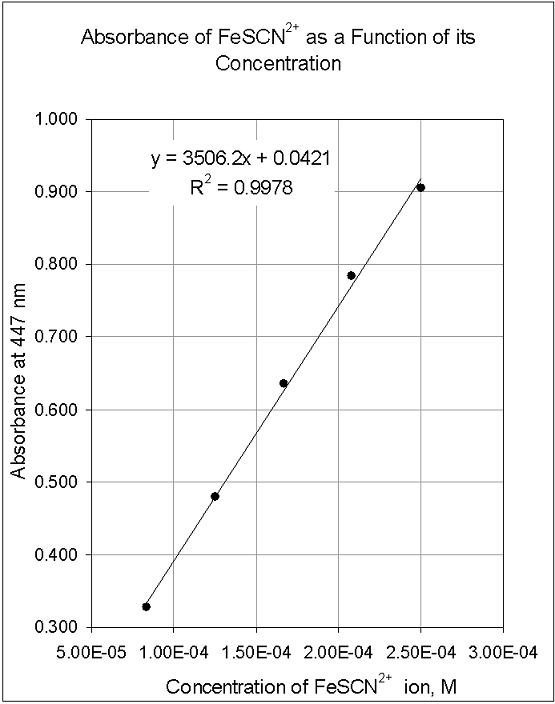
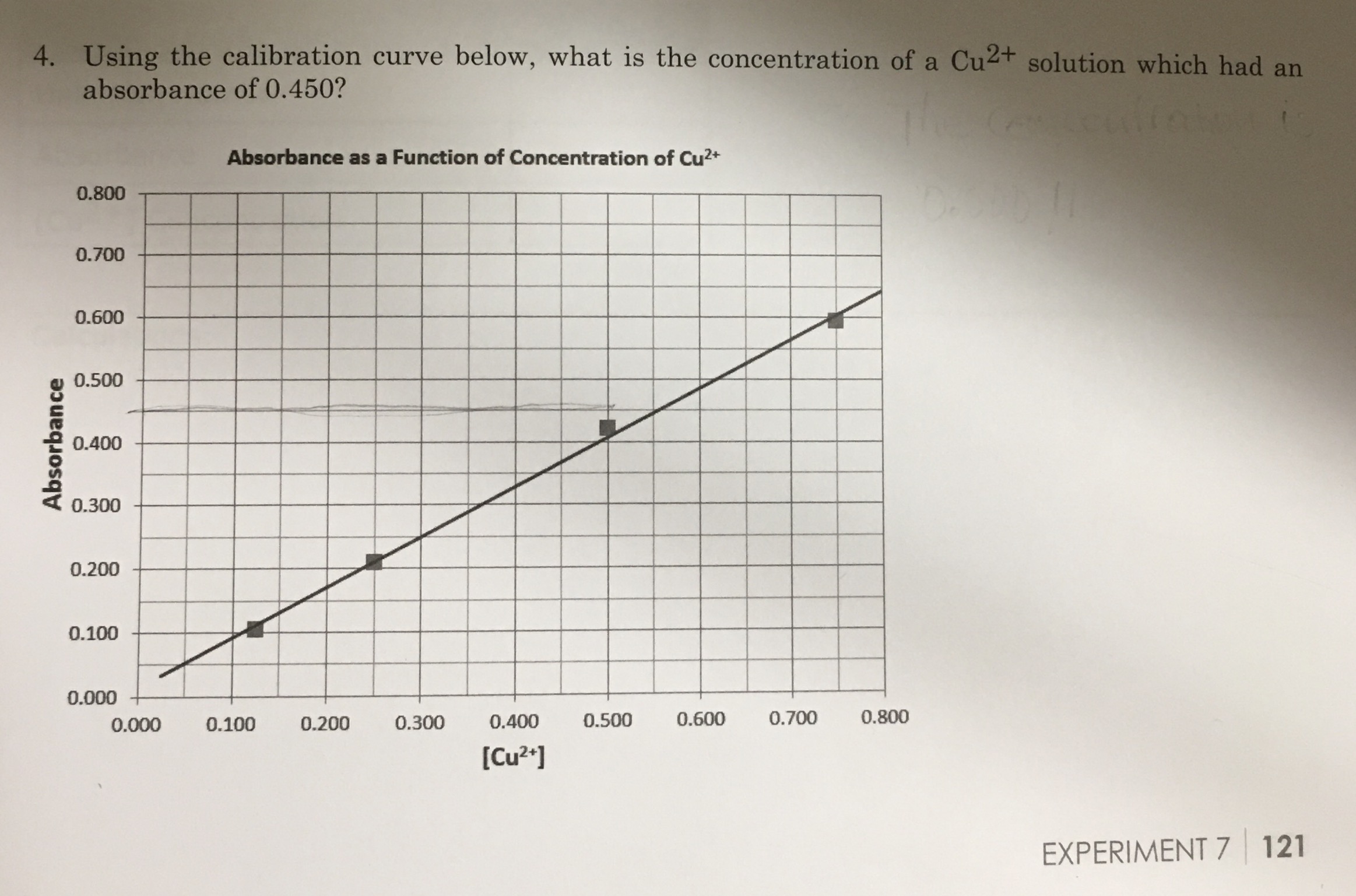
How to Draw a Standard Calibration Curve on Graph Paper & Determination of Unknown Concentration - YouTube
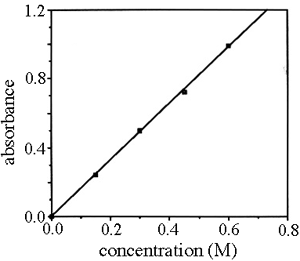
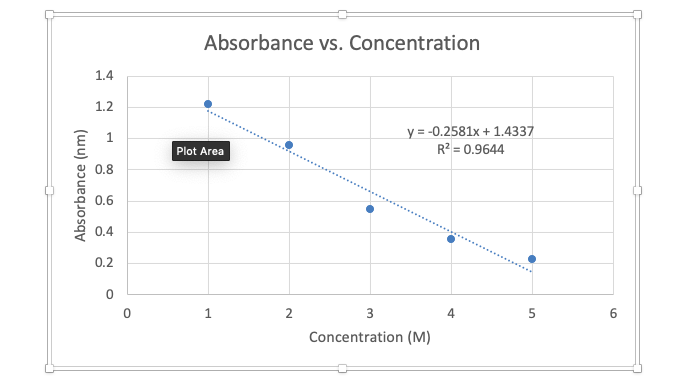
![Plot the following data as absorbance versus [ M n + ] as acalibration curve: Molar Concentration of Mn+ Absorbance,A 3.0x10-4 mol/L 0.045 6.2x10-4 mol/L 0.097 9.0x10-4 mol/L 0.14 2.2x10-3 mol/L 0. Plot the following data as absorbance versus [ M n + ] as acalibration curve: Molar Concentration of Mn+ Absorbance,A 3.0x10-4 mol/L 0.045 6.2x10-4 mol/L 0.097 9.0x10-4 mol/L 0.14 2.2x10-3 mol/L 0.](https://homework.study.com/cimages/multimages/16/calibration_curve2322129891290699101.png)
Plot the following data as absorbance versus [ M n + ] as acalibration curve: Molar Concentration of Mn+ Absorbance,A 3.0x10-4 mol/L 0.045 6.2x10-4 mol/L 0.097 9.0x10-4 mol/L 0.14 2.2x10-3 mol/L 0.
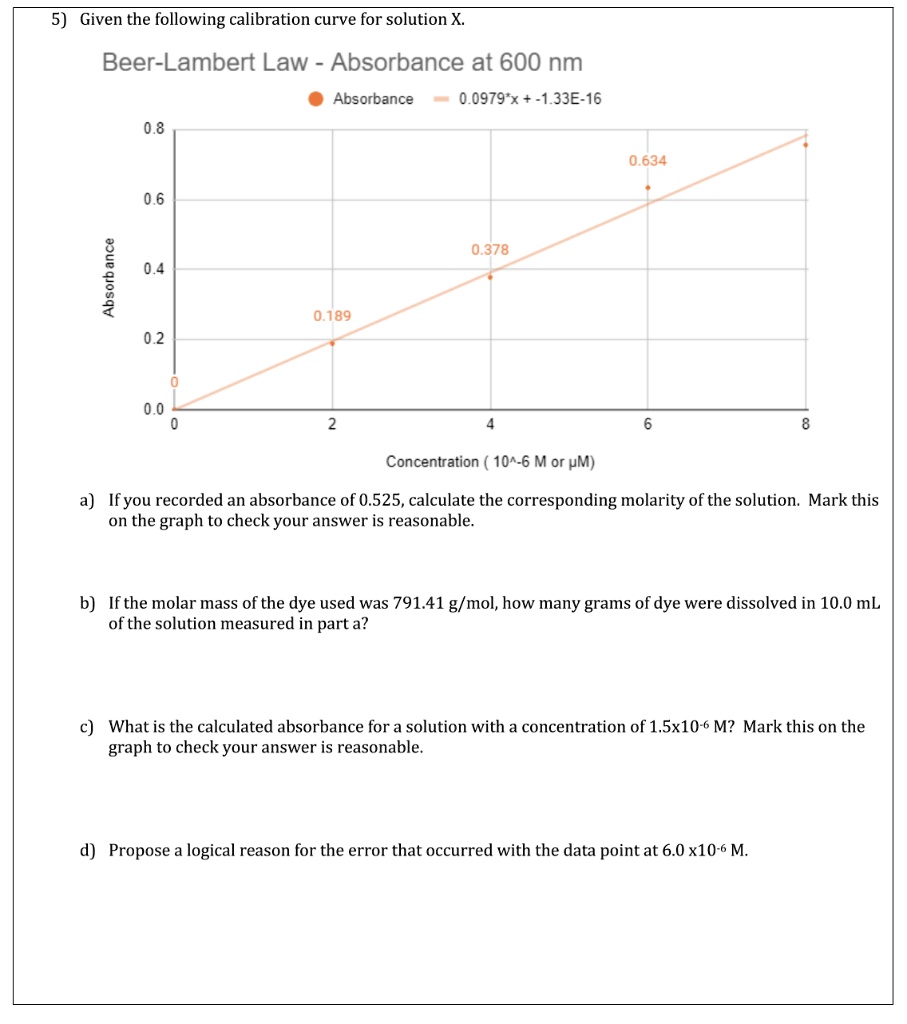
SOLVED: Given the following calibration curve for solution X Beer-Lambert Law Absorbance at 600 nm Absorbance 0979*x 1.1.33E-16 0.8 0,634 0,6 0,.378 0.4 1 0.2 0.189 Concentration ( 10^-6 M or pM)