
What is the structure of the precipitate that forms after sulfuric acid has been added to the mixture of p-aminobenzoic acid in ethanol? Why do you need to add more acid even

SOLVED: Calculate ΔGrxn at 298 K under the conditions shown below for the following reaction. SO3(g) + H2O(g) → H2SO4(l) ΔG°= -90.5 kJ P(SO3) = 0.25 atm, P(H2O) = 0.85 atm +86.7

1.) What is the structure of the precipiatate that forms after sulfuric acid has been added to the mixture of p-aminobenzoic acid in ethanol? Why do you need to add more acid

Spectroscopic Measurement of pH in Aqueous Sulfuric Acid and Ammonia from Sub- to Supercritical Conditions | Industrial & Engineering Chemistry Research
![p-Anisaldehyde (contains Acetic Acid, H2SO4) Ethanol Solution [for TLC Stain], TCI America, Quantity: 100 mL | Fisher Scientific p-Anisaldehyde (contains Acetic Acid, H2SO4) Ethanol Solution [for TLC Stain], TCI America, Quantity: 100 mL | Fisher Scientific](https://assets.fishersci.com/TFS-Assets/CCG/Chemical-Structures/chemical-structure-cas-123-11-5.jpg-650.jpg)
p-Anisaldehyde (contains Acetic Acid, H2SO4) Ethanol Solution [for TLC Stain], TCI America, Quantity: 100 mL | Fisher Scientific
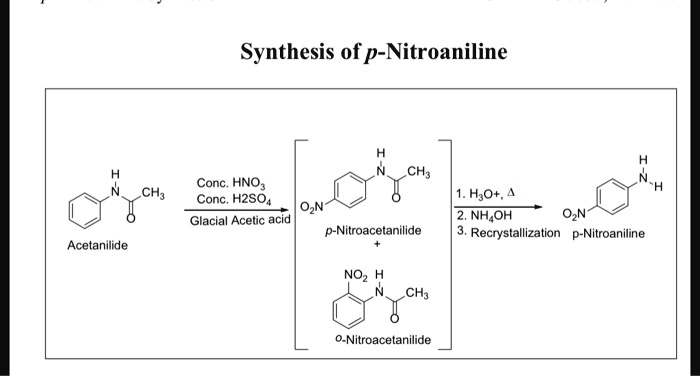
SOLVED: Synthesis of p-Nitroaniline CH; Conc. HNO3 Conc;, H2SO4 OzN Glacial Acetic acid p-Nitroacetanilide CH, HyO+, 2, NHAOH OzN- Recrystallizalion p-Nitroaniline Acetanilide NOz CH; 0-Nitroacetanilide

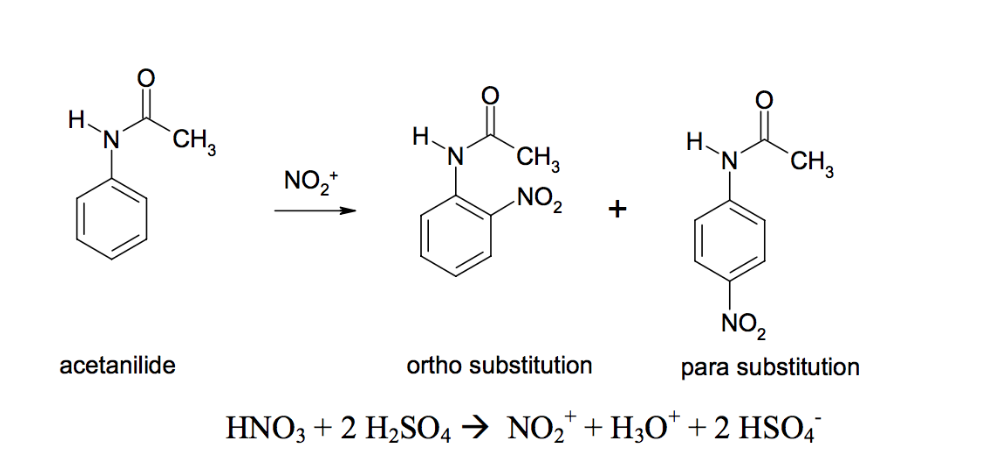







![Propyne []HgSO4 + H2SO4(1%) P Identify the product P. Propyne []HgSO4 + H2SO4(1%) P Identify the product P.](https://dwes9vv9u0550.cloudfront.net/images/1112495/0458b575-c63e-4009-82b5-2bf0590f3e45.jpg)

