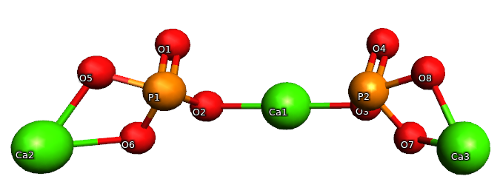
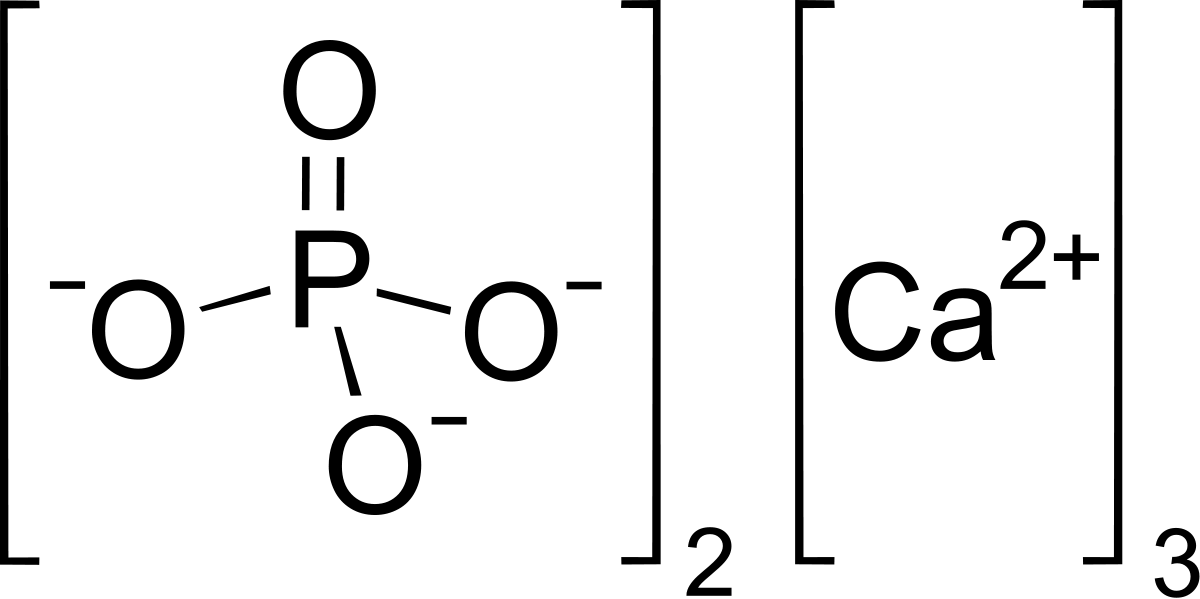
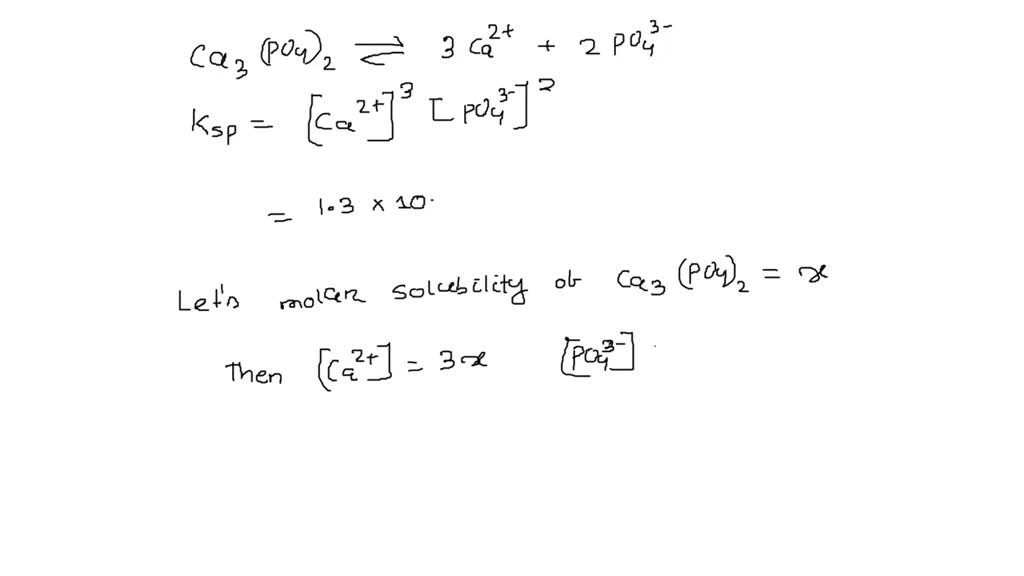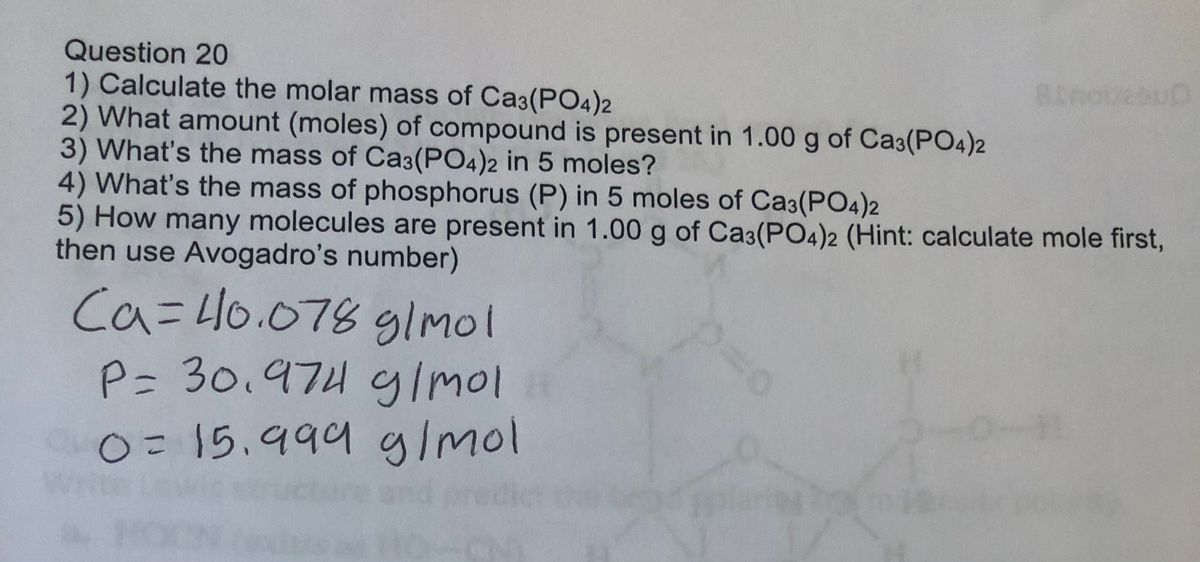How to Balance Ca3(PO4)2 + SiO2 + C = CaSiO3 + P4 + CO | How to Balance Ca3(PO4)2 + SiO2 + C = CaSiO3 + P4 + CO Are you ready

Formation of discrete β-Ca3(PO4)2-Y2O3 phase mixtures influenced through elevated heat treatments - ScienceDirect
![Amazon.com: Calcium Phosphate [Ca3(PO4)2] 99.9% ACS Grade Powder 1.5 Lb in Three Space-Saver Bottles USA : Health & Household Amazon.com: Calcium Phosphate [Ca3(PO4)2] 99.9% ACS Grade Powder 1.5 Lb in Three Space-Saver Bottles USA : Health & Household](https://m.media-amazon.com/images/I/91+-vBAT3dL._AC_UF1000,1000_QL80_.jpg)
Amazon.com: Calcium Phosphate [Ca3(PO4)2] 99.9% ACS Grade Powder 1.5 Lb in Three Space-Saver Bottles USA : Health & Household
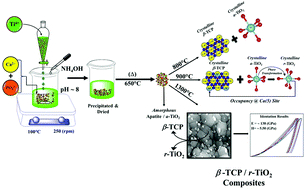
Sequential elucidation of the β-Ca3(PO4)2/TiO2 composite development from the solution precursors - Dalton Transactions (RSC Publishing)
![aasign oxidation number to the element ca3[PO4]2 - Chemistry - Redox reactions - 16636031 | Meritnation.com aasign oxidation number to the element ca3[PO4]2 - Chemistry - Redox reactions - 16636031 | Meritnation.com](https://s3mn.mnimgs.com/img/shared/content_ck_images/ck_608a5f315ed80.jpeg)
aasign oxidation number to the element ca3[PO4]2 - Chemistry - Redox reactions - 16636031 | Meritnation.com

Phase and melting relationships of β, α and α′-Ca3(PO4)2 polymorphs in the Ca3(PO4)2-Zn3(PO4)2 system - ScienceDirect