
Articulatory Underpinnings of Reduced Acoustic–Phonetic Contrasts in Individuals With Amyotrophic Lateral Sclerosis | American Journal of Speech-Language Pathology
Open-File Report 94-603 This report is preliminary and has not been reviewed for conformity with U.S. Geological Survey editoria
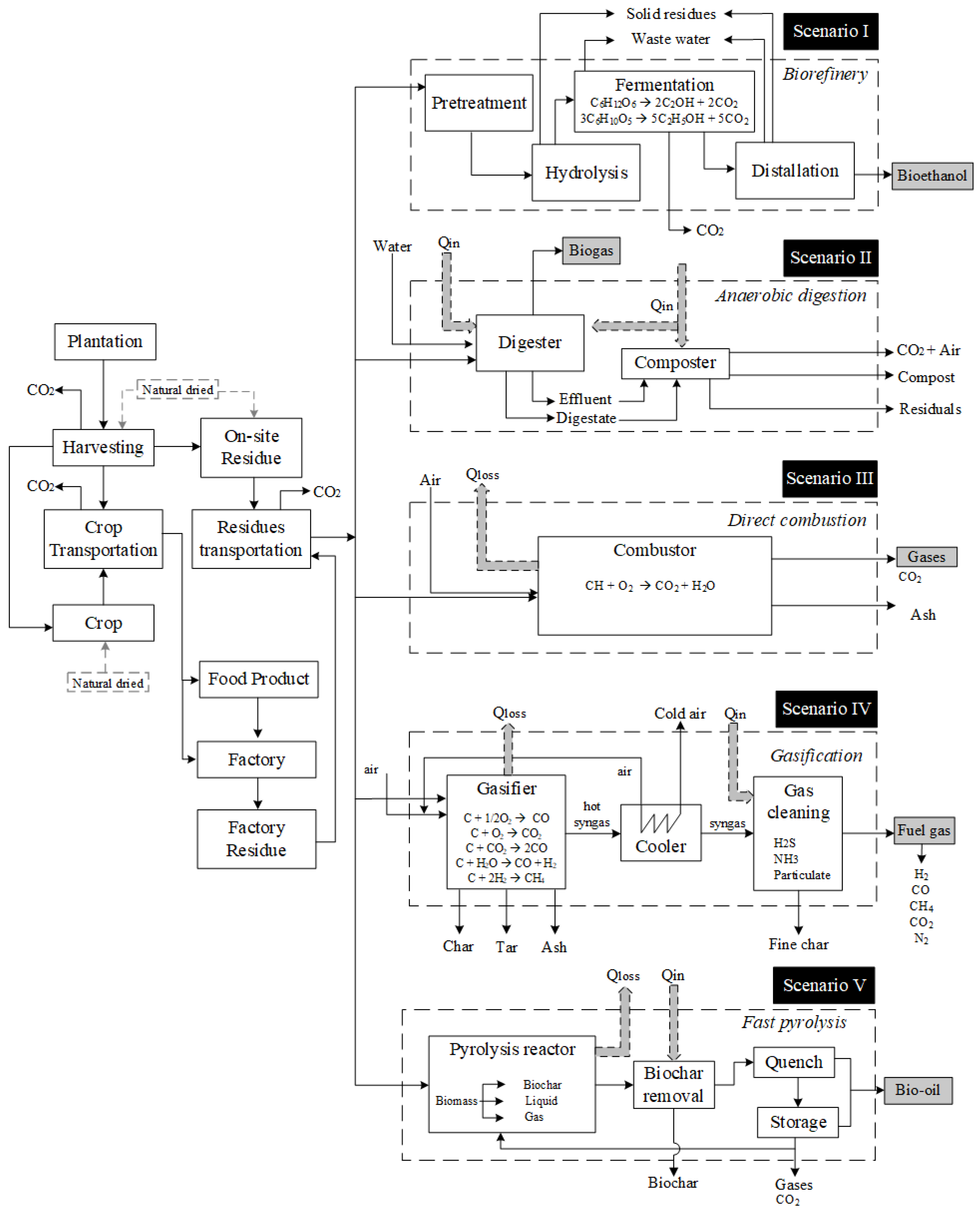
Energies | Free Full-Text | Renewable Energy Potential and CO2 Performance of Main Biomasses Used in Brazil
![Synthesis and Structure−Activity-Relationships of 1H-Imidazo[4,5-c]quinolines That Induce Interferon Production | Journal of Medicinal Chemistry Synthesis and Structure−Activity-Relationships of 1H-Imidazo[4,5-c]quinolines That Induce Interferon Production | Journal of Medicinal Chemistry](https://pubs.acs.org/cms/10.1021/jm049211v/asset/images/medium/jm049211vf00002.gif)
Synthesis and Structure−Activity-Relationships of 1H-Imidazo[4,5-c]quinolines That Induce Interferon Production | Journal of Medicinal Chemistry
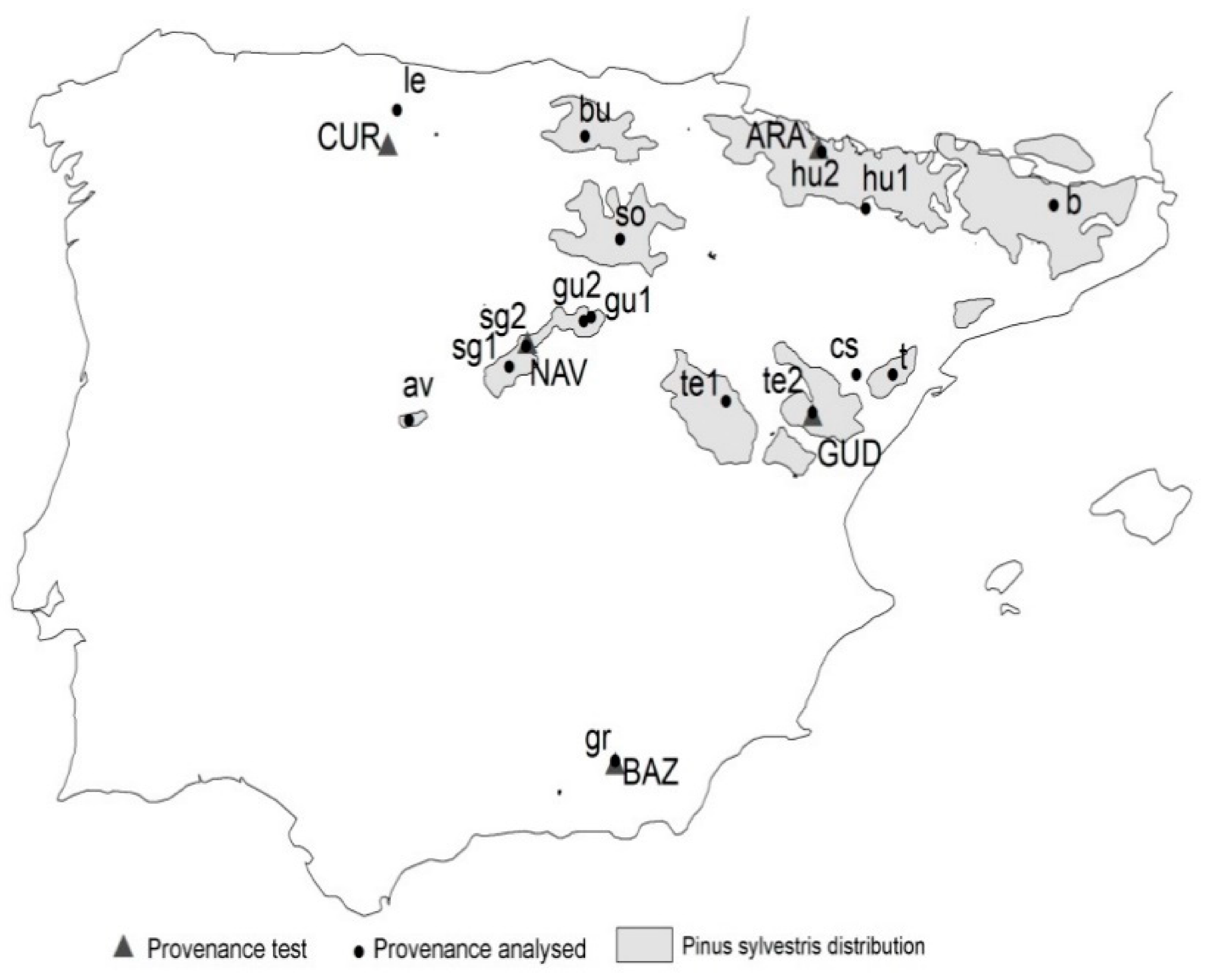
Forests | Free Full-Text | Seed Sourcing Strategies Considering Climate Change Forecasts: A Practical Test in Scots Pine

Atmosphere | Free Full-Text | Future Projections for Wind, Wind Shear and Helicity in the Iberian Peninsula

Discovery of CRBN E3 Ligase Modulator CC-92480 for the Treatment of Relapsed and Refractory Multiple Myeloma | Journal of Medicinal Chemistry

Deconstructing Noncovalent Kelch-like ECH-Associated Protein 1 (Keap1) Inhibitors into Fragments to Reconstruct New Potent Compounds | Journal of Medicinal Chemistry

Dibenzodiazepinone-type muscarinic receptor antagonists conjugated to basic peptides: Impact of the linker moiety and unnatural amino acids on M2R selectivity - ScienceDirect
![Compounds useful for inhibiting ROR.gamma.t Patent Grant Clarke , et al. May 18, 2 [Eli Lilly and Company] Compounds useful for inhibiting ROR.gamma.t Patent Grant Clarke , et al. May 18, 2 [Eli Lilly and Company]](https://uspto.report/patent/grant/11008336/US11008336-20210518-C00001.png)
Compounds useful for inhibiting ROR.gamma.t Patent Grant Clarke , et al. May 18, 2 [Eli Lilly and Company]

Total Synthesis of a Mycobactin S, a Siderophore and Growth Promoter of Mycobacterium Smegmatis, and Determination of its Growth Inhibitory Activity against Mycobacterium tuberculosis | Journal of the American Chemical Society

Discovery, Structure−Activity Relationship, and Pharmacological Evaluation of (5-Substituted-pyrrolidinyl-2-carbonyl)-2-cyanopyrrolidines as Potent Dipeptidyl Peptidase IV Inhibitors | Journal of Medicinal Chemistry









